the2020s
2021
October 1st
SUMITOMO BAKELITE CO., LTD. and KAWASUMI LABORATORIES, INC.,
integrated our medical device business, and start "SB-KAWASUMI LABORATORIES, INC." as the brand new company.

1st Oct 2021 SB-KAWASUMI LABORATORIES, INC.
2021
Started Production/sales of "Heart Positioner for Off-Pump CABG" in the United States.
2020
Became a group company of Sumitomo Bakelite Co., Ltd. (First Section of the Tokyo Stock Exchange)
Delisted from the Second Section of the Tokyo Stock Exchange.
2020
Obtained Japanese Medical Device Approval of "Esophageal Stent".
the2010s
2018
Started production/sale of "biliary stent".

2017
Succeeded to the Manufacture and Sales rights of "Low-Pressure Drainage Unit" and "Billially Stent".
Started sales of "Monopolar ESD Knife" in the United States.
2015
Started production/sale of "Plasma Separator".
2015
Concluded the exclusive distribution agreement for "Steerable Microcatheter" (Overseas product name : SwiftNINJA) with the US distributor.
2014
Opened European office at Sumitomo Bakelite Europe N.V., Expanded sales in European Market.
Acquired Vaupel Holdings, Inc. in the United States.
2013
Started production/sale of "Aortic Aneurysm Stent Graft".
2013
Expanded the domestic sales branches.
2010
Started production/sale of "Polysulfone membrane dialyzers".
2010
Obtained Japanese Medical Device Approval of "Low-Pressure Suction Drain Bag".
the2000s
2009
Obtained trade rights of Therapeutic Apheresis by transfer.

2009
Opened Chinese office in Shanghai. Entered the Chinese market.
Obtained Japanese Medical Device Approval of "Monopolar ESD Knife".

2005
Launched the Medical device brand " sumius".
2004
Started production/sale of "blood component collection systems with leukocyte reduction filters".
2002
Started production/sales of "potassium adsorption filter".

2002
Opened Plant in Dongguan (Guangdong Province), Started manufacture of Medical Devices in China.

2001
Started overseas production/sale of "blood bags with leukocyte reduction filters".
the1990s
1999
Constructed Korat Plant of KAWASUMI LABORATORIES (THAILAND)CO., LTD. in Thailand.
1999
Obtained Japanese Medical Device Approval of "Percutaneous Transesophageal Gastrostomy kit" .
1995
Started production/sale of "Apheresis kits".
1994
Obtained Japanese Medical Device Approval of "Flexible Overtube".
Started sale of "Pneumo-Activate EVL Device".
1992
Started production/sale of "Blood Bag with New Additive MAP Solution".
1991
Established KAWASUMI LABORATORIES AMERICA, INC. in USA.
1990
Four subsidiaries in Akita area were merged to form Akita Sumitomo Bakelite Co., Ltd.

Sumibe Medicalcare Co., Ltd. was established for maintenance of homecare medical equipment.
the1980s
1989
Obtained Japanese Medical Device Approval of "Balloon Catheter for vascular treatment" (Aortic occlusion Balloon Catheter).
1987
Listed in the Second Section of the Tokyo Stock Exchange.
1986
Sumibe Medical Co., Ltd. was established for manufacture and research of Medical devices.
1984
Obtained Japanese Medical Device Approval of " Portable Low-Pressure Suction Drain" and "Thoracic Drainage Bag Connectable to Suction Pumps".
1983
Obtained Japanese Medical Device Approval of "Endotracheal Suction Kit" and " Urinary Drainage Bag".
1980
Japan Red Cross started to use our Blood bag.
the1970s
1979
Medical Device Laboratory was opened in the Central Research Laboratory.
1978
Established a joint venture, THAI KAWASUMI CO., LTD. in Thailand(present name : KAWASUMI LABORATORIES (THAILAND) CO., LTD.).
1977
Developed Blood bags.
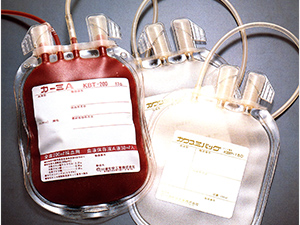
1976
Set up the Medical Research Group in the Central Research Laboratory.
1975
Set up the Medical Device development Department.
Started the medical device products business.
1973
Developed the first Japanese Coil-type dialyzer"DC-7".
Winner of award of Japan Society of Plastics Technology at 15th Japan Plastics product competition.

the1960s
1969
Started production/sale of "Blood tubing lines" for hemodialysis.
1967
Constructed Saiki Plant in Oita pref., Japan.
1962
Central Research Laboratory was opened.
(Put on only the history of Medical Device Division.)
the1950s
1957
Established KAWASUMI LABORATORIES, INC. in Tokyo, Japan.

1955
Founded Sumitomo Bakelite Co., Ltd.
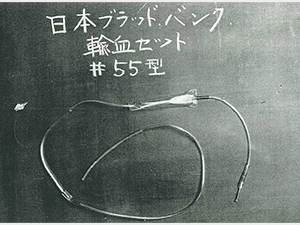
1954
Started production of the first Japanese disposable plastic blood collection/transfusion sets.